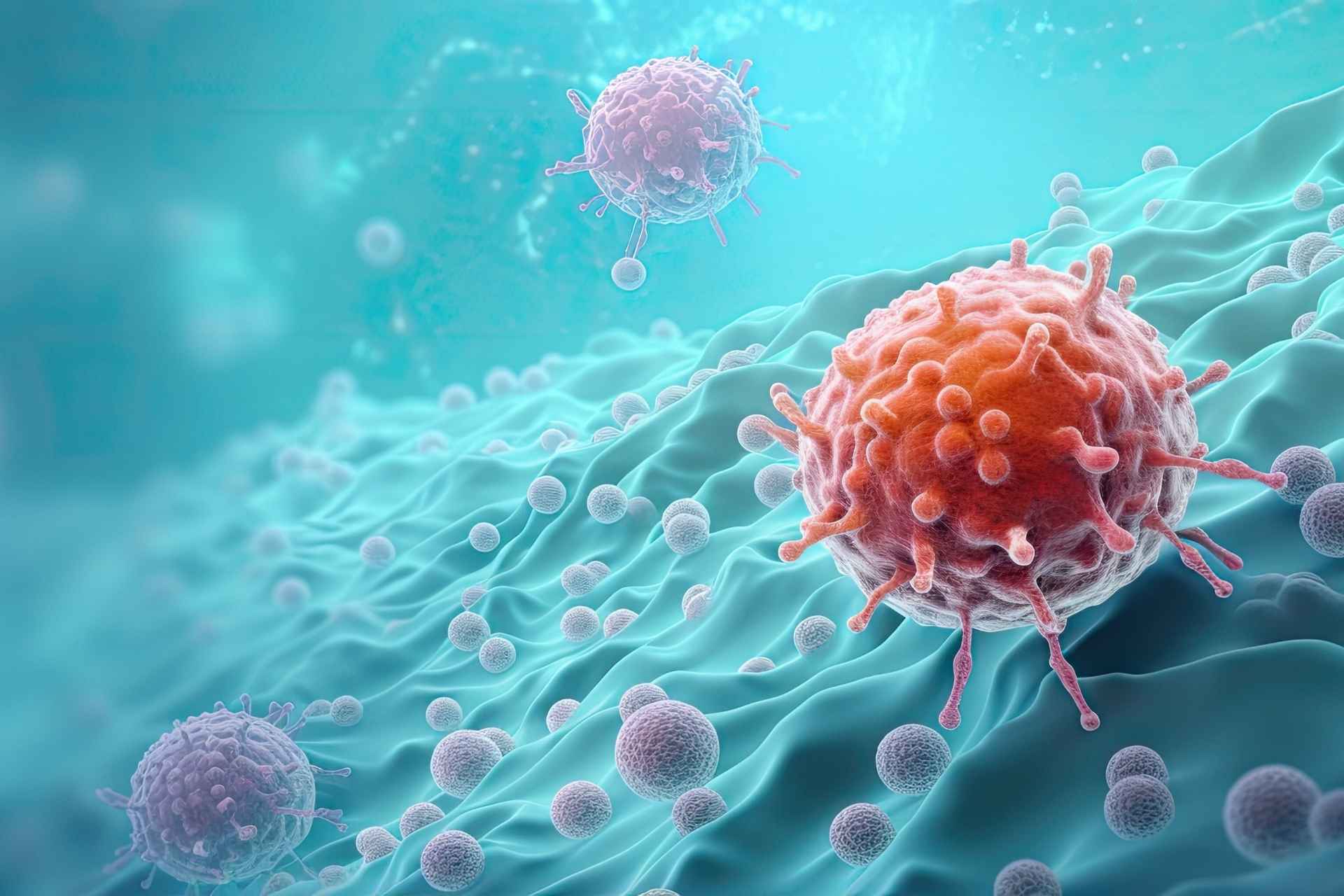
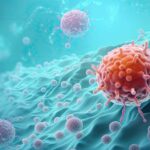
Gut microbes have been linked to cancer development, progression and responses to treatment. Now, researchers have found that a single gut bacterial gene can control how dietary nutrients are shared between tumors and immune cells—at least in mice.
The findings, published in Cell Host & Microbe, suggest that specific microbial genes can affect how diet affects cancer progression.
Scientists have known that gut bacteria consume nutrients and produce metabolites that shape host physiology, and that dietary supplements can have mixed or even opposite effects in different individuals. However, it’s not known how specific microbial genes control nutrient availability inside tumors, nor how these genes interact with diet to influence immune cells and tumor outcomes.
Working in mice, Shanshan Qiao at Cornell University in New York and her colleagues tested whether gut bacteria change how dietary amino acids influence cancer growth.
Promoting cancer
The researchers compared mice with gut microbes to mice that lacked them and fed both groups diets with either low or high amino acid content. In mice without gut microbes, diets rich in amino acids led to higher amino acid levels in the body and faster tumor growth, but this effect was much weaker in mice with normal gut bacteria.
Human gut bacteria vary widely in how much they consume amino acids, and when these bacteria were transferred into mice, those receiving microbes that consumed more amino acids ended up with lower amino acid levels in their bodies. These animals also developed smaller tumors.
Next, the researchers studied individual bacterial genes that break down amino acids in the gut and found that when these genes were missing, more amino acids remained available in the body and within tumors, promoting their growth.
Precision medicine
The team focused on a bacterial gene, called bo-ansB, that breaks down the amino acid asparagine. When bacteria were able to use asparagine, amino acid supplements worsened tumors, but when the bacteria could not use asparagine, the supplements improved tumor control by supporting cancer-fighting immune cells.
When bo-ansB was removed, more asparagine reached tumors, boosting immune responses and improving the effectiveness of anti-cancer therapy, the researchers found.
The findings suggest that gut microbes can influence cancer outcomes by regulating nutrient availability and uptake in immune cells, the authors say. The results, they add, “establish the gut microbiome as a genetically tractable ‘metabolic organ’ and uncover a regulatory layer linking dietary amino acid, microbiota function, and cancer—offering opportunities for precision medicine.”