• Sugar control
• Side effects
What is already known on this topic
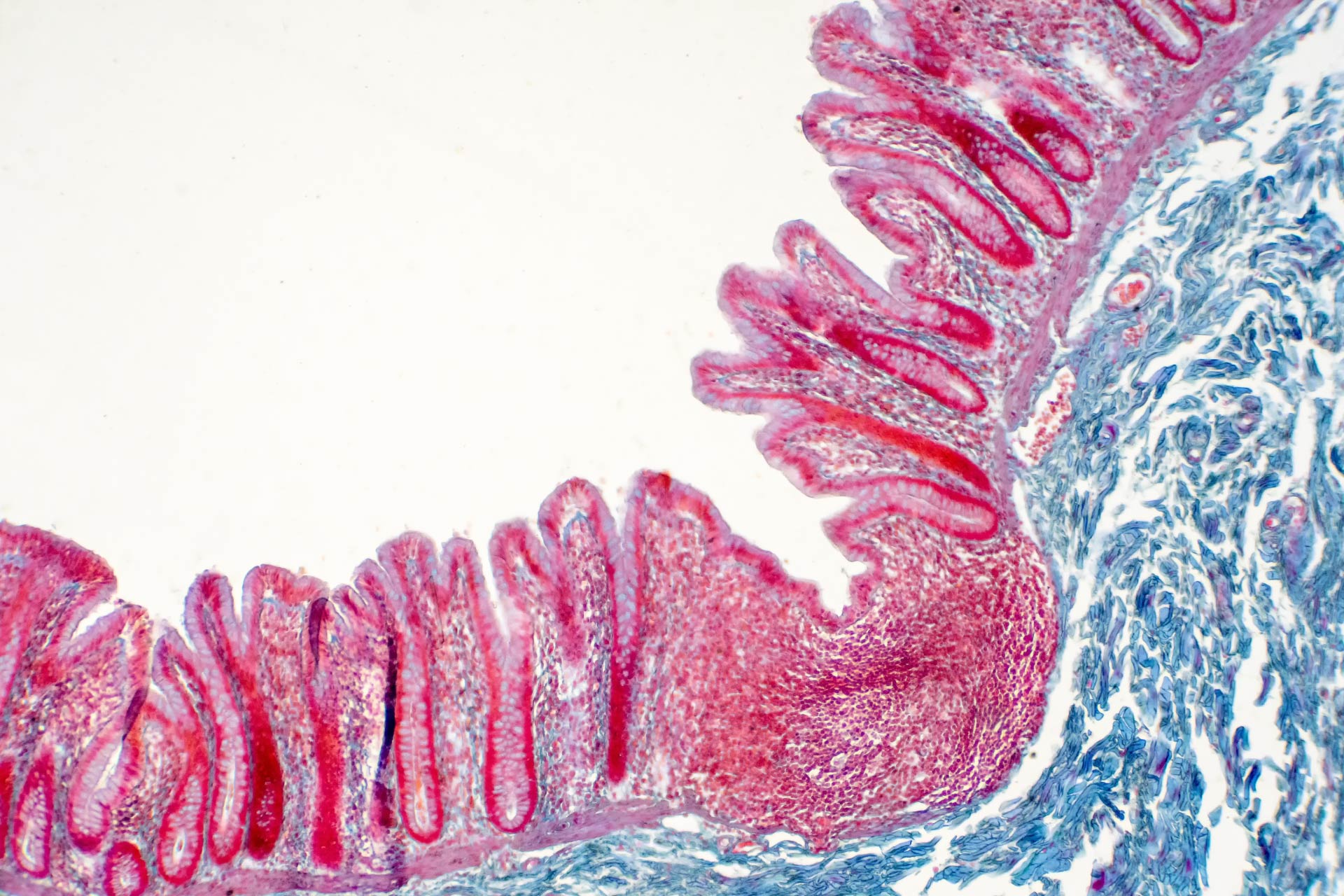
The enteric nervous system consists of a mesh-like system of neurons that govern the function of the gastrointestinal tract. Several studies have shown that the gut microbiota modulates the development and function of the enteric nervous system, and alterations in the composition of the gut microbial community can lead to metabolic disorders such as obesity and diabetes. But the molecular pathways that mediate the communication between the gut and enteric neurons remain poorly understood.What this research adds
Working in mice, researchers discovered a population of enteric neurons called CART+, which reside in the gastrointestinal regions where most microorganisms are found. CART+ neurons, which are modulated by the gut microbiota, help to regulate blood sugar levels, insulin and feeding behavior. Removing the microbiota caused the loss of CART+ neurons and impaired glucose regulation, which in turn can lead to metabolic diseases such as type 2 diabetes.Conclusion
The findings suggest that targeting these types of neural pathways could ease the adverse effects on the central nervous system that are common among metabolic disorder treatments.
Often referred to as “the second brain”, the enteric nervous system consists of a mesh-like system of neurons that govern the function of the gastrointestinal tract. Now researchers have found that a gut microbiota-modulated neural pathway can regulate blood sugar independently from the central nervous system.
The findings, published in Science, suggest that targeting these types of neural pathways could ease the adverse effects on the central nervous system that are common among metabolic disorder treatments.
Several studies have shown that the gut microbiota modulates the development and function of the enteric nervous system, and alterations in the composition of the gut microbial community can lead to metabolic disorders such as obesity and diabetes. But the molecular pathways that mediate the communication between the gut and enteric neurons remain poorly understood.
To better characterize the role of the enteric nervous system, Paul Muller and Daniel Mucida at The Rockefeller University and their colleagues set out to determine how the gut microbiota impacts enteric neurons.
Sugar control
Working in mice, the researchers discovered a population of enteric neurons called CART+, which reside in the ileum and colon—the gastrointestinal regions where most microorganisms are found.
The team discovered that CART+ neurons, which are modulated by the gut microbiota, help to regulate blood sugar levels, insulin and feeding behavior through neural pathways to the liver and pancreas.
Removing the microbiota caused the loss of CART+ neurons and impaired glucose regulation, independently from the central nervous system. Impaired regulation of blood sugar can in turn lead to metabolic diseases such as type 2 diabetes.
Side effects
The results revealed a microbial-dependent function of enteric neurons, which have the potential to perform metabolic control independent of the central nervous system. Further experiments showed that molecules typically linked to inflammation play a key role in controlling the number of enteric neurons and the regulation of glucose levels in response to the microbiota.
It’s still unclear whether CART+ neurons respond to the presence of glucose in the gut, release of neurotransmitters, or the movement of fecal matter, the researchers say. But the findings suggest that targeting neural circuits such as the one uncovered in the study could help to bypass the undesirable effects on the central nervous system that are often associated with the treatment of metabolic disorders such as type 2 diabetes, the authors add.