What is already known
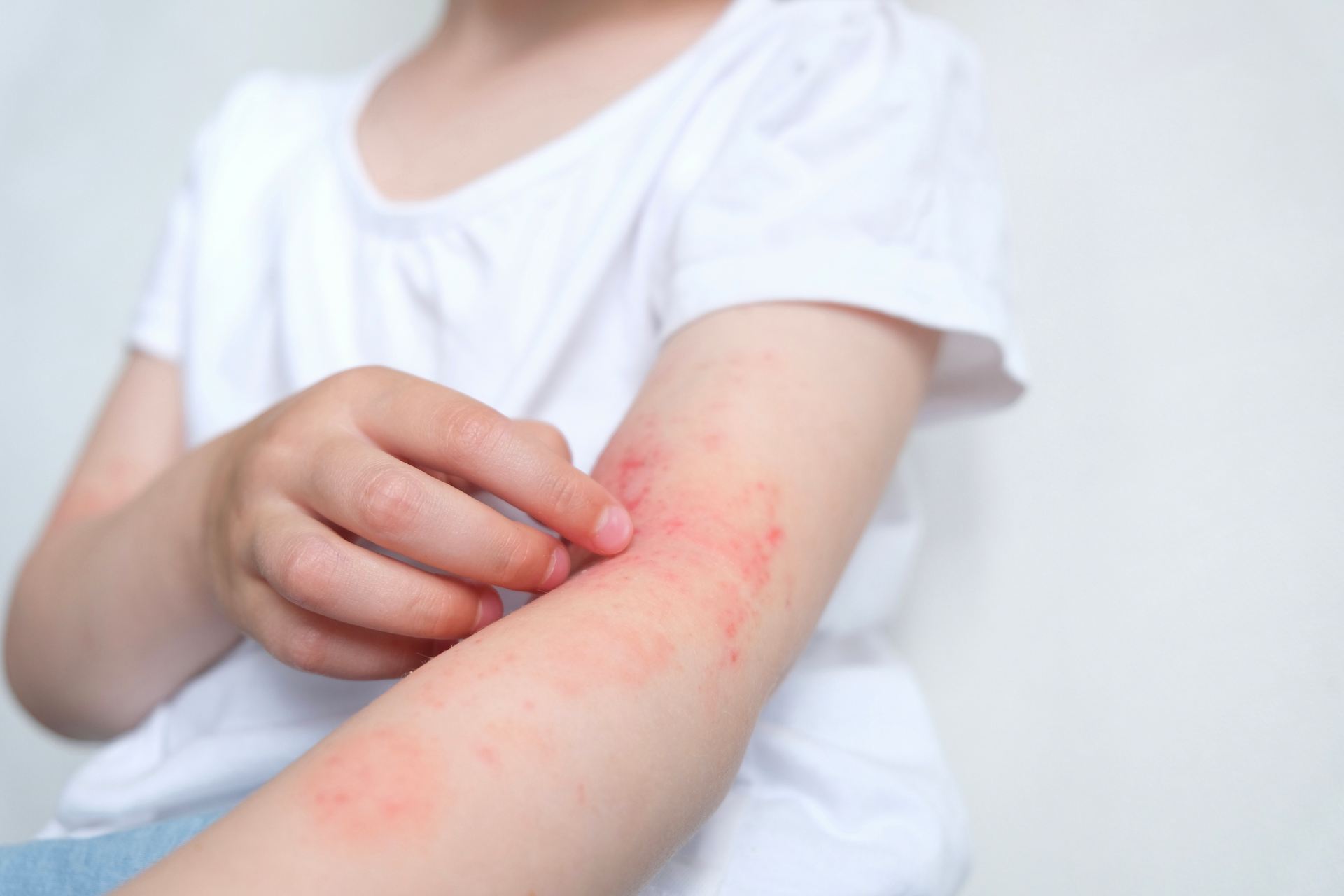
Atopic dermatitis (AD) is a chronic inflammatory skin disease that affects up to 20% of people and is particularly common in children. Staphylococcus aureus, an opportunistic pathogen, often inhabits the inflamed skin of individuals with AD, exacerbating the disease by damaging the skin. However, specific mutations and the impact of de novo microbiome mutations on complex diseases has not been fully understood.
What this research adds
By monitoring 23 children receiving treatment for AD, researchers discovered that S. aureus changes through acquisition of new mutations during skin colonization. Every patient had a primary S. aureus population dominated by a single lineage, with occasional infiltration from other groups. Over several months, some variants spread throughout the body with signs of adaptive evolution. One notable example was the parallel evolution of mutations in the capD gene, responsible for capsule synthesis, in one patient and across-body spread in two patients, and capD negativity in S. aureus was found more prevalent in AD than other conditions.
Conclusions
The study highlights the importance of evaluating microbes’ mutations in understanding the contribution of microbes to complex diseases.
One common occurrence in symptomatic AD skin is colonization by the opportunistic pathogen S. aureus, which is usually only found in low amounts on healthy skin. These bacteria cause various human infections and is believed to originate from the nares of healthy individuals. Different S. aureus sequence types have varying virulence potential, antibiotic resistance, and disease contexts. However, no sequence types have been found to be strongly associated with AD. While de novo mutations in S. aureus have been observed in children with AD, their fate and consequences are unknown.
Tami D. Lieberman and colleagues, recently published a prospective longitudinal study in Cell Host & Microbe Journal, where they monitored S. aureus skin colonization and its evolution in 23 children from Mexico aged between 5-15 years old who had moderate to severe AD.
Researchers found out that S. aureus quickly adjusts on people undergoing AD treatment, by developing de novo mutations. These adaptive mutants spread throughout the body, including the nares, and individuals with AD are more likely to have loss-of-function mutations in the capD gene of S. aureus, which is closely linked to its virulence, compared to healthy individuals.
Longitudinal tracking of S. aureus evolution in AD patients
Swabs were collected from affected and unaffected skin sites of each child, and culture-based whole-genome sequencing was performed to detect mutations acquired by S. aureus as a result of natural selection in individual people. By clustering each patient’s isolates into lineages, researchers found that most patients were colonized by a single major lineage, although minor lineages were recovered in some people.
S. aureus mutants sweep across the body
Visualizing the temporal evolutionary dynamics, it was observed that S. aureus accumulated mutations over time and genotypes with newly acquired mutations repeatedly replaced the existing diversity. Interestingly, the spread of new mutations was not spatially restricted but spanned the entire body, including unaffected skin and the natural habitat of the nose. These data suggesed that new S. aureus genotypes commonly emerge via on-person de novo mutation in people being treated for AD, some of which subsequently sweep the on-person population.
Adaptive mutations alter polysaccharide capsule
To test whether the de novo mutations provided a competitive advantage on AD skin, researchers investigated mutated genes for adaptive signatures. Observations of parallel evolution within a patient, parallel evolution across patients, and association with replacement events suggested that alterations in capD provide a survival advantage for S. aureus on AD skin. The capD gene encodes an enzyme involved in the first step of capsular polysaccharide synthesis, a virulence factor that protects the pathogen from phagocytosis and the innate immune system. No other adaptive de novo mutations in S. aureus genes other than capD were observed.
Capsule loss is common in AD globally
The occurrence of capD-truncation mutations on AD skin indicated that having an acapsular phenotype may provide an advantage on AD skin. Selective pressures for capsule loss could include immune escape from capsule-targeted antibodies or innate immune components, increased adherence to AD skin, and alleviation of metabolic costs. The latter may explain the preponderance of mutations in the first gene in the biosynthetic pathway, capD, rather than an even distribution across all capsule-associated genes.
All together, these findings emphasize how de novo mutations can influence the competitive ability of bacteria in skin colonization and how mutation tracking is useful in discovering possible therapeutic avenues. Further studies will be required to characterize the mechanistic basis behind the CapD-negative advantage on AD skin in order to design target therapies that lower the burden of S. aureus colonization in AD.