• The lectin receptor Mincle senses gut commensals
• Mincle triggers cytokine production in response to gut microbes
• Mincle contributes to intestinal barrier function
-
What is already known on this topic
The intestinal barrier is important for keeping gut microbes in check and avoid their spread to other sites of the body, which can result in inflammation. But how gut cells sense commensal bacteria is poorly understood. -
What this research adds
Immune cells in the gut sense commensal microbes through specific receptors that trigger the production of several inflammatory molecules, enhancing the function of the intestinal barrier. -
Conclusions
The findings shed light on a new mechanism of interaction between the host and gut commensals, which has potential implications for disorders associated with increased bacterial translocation.
Cells in the gut sense commensal microbes through specific receptors that trigger the production of several inflammatory molecules and the activation of the immune system. This enhances the function of the gut barrier and prevents microbes from leaving the gut and causing inflammation to other sites of the body, researchers report in the journal Immunity. The study, done in mice, was led by María Martínez-López at the Spanish National Center for Cardiovascular Research in Madrid.
Gut microbes are critical for host metabolism and immune system development, but they have to be kept in check to avoid their migration to other sites in the body, where they can cause inflammation.
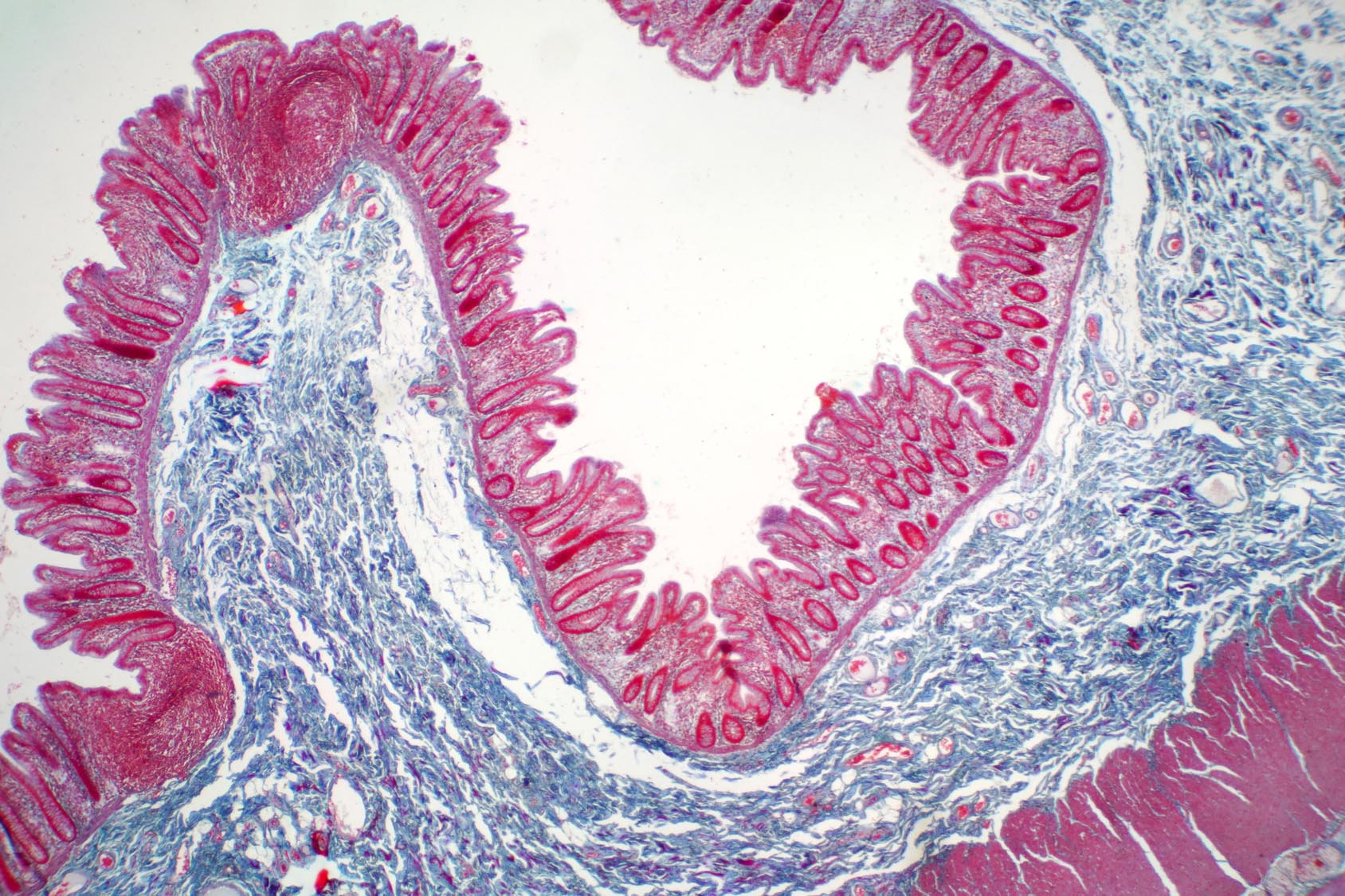
The cells lining the intestine, the gut’s mucus layer, antimicrobial peptides, and microbe-specific immunoglobulins make up the intestinal barrier, which ensures that gut microbes do not leave the gut. This barrier is regulated by immune cells including T helper 17 (Th17) cells.
In the gut, specific receptors that bind to lectin molecules are able to recognize components of the cell wall of fungal and bacterial pathogens, stimulating the activation of Th17 cells and triggering the production of several inflammatory molecules known as cytokines.
For this reason, the researchers set out to assess whether some of these lectin receptors could help to sense commensal bacteria and promote immune cell function in the gut.
The lectin receptor Mincle senses gut commensals
In experiments done in a laboratory dish, gut microbes stimulated the activation of Th17 cells only when intestinal immune cells expressed a specific lectin receptor called Mincle.
In following experiments done in mice, the researchers found that the lectin receptor Mincle could bind some families of gut bacteria, including Lactobacillus.
Mincle triggers cytokine production in response to gut microbes
Bacteria such as Lactobacillus tend to bind to the lining of small masses of lymphatic tissue in the small intestine known as Peyer’s patches. For this reason, the team hypothesized that Mincle could be expressed by cells located in these masses of lymphatic tissue. Indeed, Mincle was expressed by immune cells within Peyer’s patches.
Next, the researchers found that, after being exposed to gut microbes, these cells produce the cytokines IL-6 and IL-23p19, but only when the receptor Mincle is present. Mice that lack Mincle produced lower levels of these molecules.
Because IL-6 and IL-23p19 regulate the production of the cytokines IL-17 and IL-22 by T cells, the researchers looked at the effects of Mincle on IL-17 and IL-22. T cells from Peyer’s patches produced high levels of IL-17 and IL-22 in mice that expressed Mincle, but not in mice that didn’t, suggesting that the receptor is needed for the production of these two cytokines in the gut.
However, gut immune cells produced IL-17 only when commensal bacteria were present. Indeed, in mice treated with broad-spectrum antibiotics, which disrupt the gut microbiota, the researchers could not detect IL-17.
Mincle contributes to intestinal barrier function
Next, the team tested whether the absence of Mincle affected the intestinal barrier function.
Mice lacking Mincle had low levels of antimicrobial peptides and IgA, and showed increased rates of microbial translocation to the liver, which resulted in liver inflammation and impaired function.
These findings suggest that Mincle fosters the intestinal barrier, preventing microbes from migrating to other body sites and causing inflammation.
The study reveals a new mechanism through which the host and its commensals interact. This mechanism, the authors say, has potential implications for metabolic disorders and other diseases associated with increased bacterial translocation.